The agency also is responsible for the safety and security of our nation’s food supply, cosmetics, dietary supplements, products that give off electronic radiation, and for regulating tobacco products. Department of Health and Human Services, protects the public health by assuring the safety, effectiveness, and security of human and veterinary drugs, vaccines and other biological products for human use, and medical devices. Plasma from people who have recovered from COVID-19 will have. It contains antibodies that help fight off infections. We encourage individuals to consider donating and hope this information will serve as a helpful resource to facilitate this important act of kindness. Plasma is the liquid portion of your blood. People who have fully recovered from COVID-19 for at least two weeks can contact their local blood or plasma collection center today to schedule an appointment. Convalescent plasma can also be used to manufacture a biological product called hyperimmune globulin, which can similarly be used to treat patients with COVID-19. In fact, one donation has the potential to help up to four patients. Those individuals who have recovered from COVID-19 could have an immediate impact in helping others who are severely ill. The American Red Cross has also set up a website for interested donors ( and the FDA continues to work with others in this area to help encourage additional donations.ĭuring this challenging time, many people are asking what they can do to contribute to the COVID-19 response.
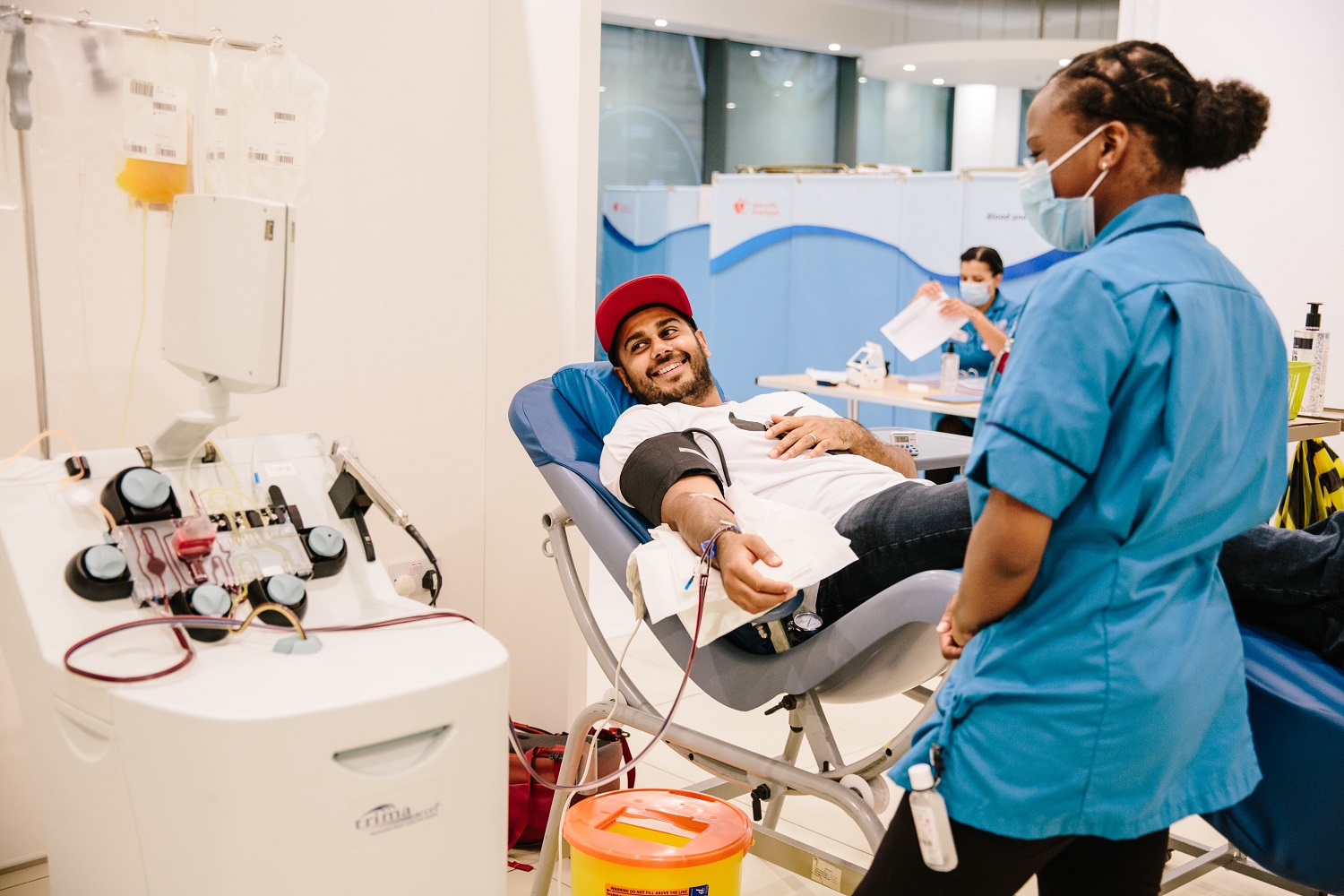
The webpage also provides information for those interested in participating in the expanded access protocol, conducting clinical trials or submitting eIND applications. The FDA has launched a new webpage to guide recovered COVID-19 patients to local blood or plasma collection centers to discuss their eligibility and potentially schedule an appointment to donate. A number of clinical trials are also taking place to evaluate the safety and efficacy of convalescent plasma and the FDA has granted numerous single patient emergency investigational new drug (eIND) applications as well.Īs this work moves forward, the key to ensuring the availability of convalescent plasma to those in greatest need is getting recovered COVID-19 patients to donate plasma. More than 1,040 sites and 950 physician investigators nationwide have signed on to participate in the Mayo Clinic-led expanded access protocol. The response to the agency’s recently announced national efforts to facilitate the development of and access to convalescent plasma has been tremendous. It is important that we evaluate this potential therapy in the context of clinical trials, through expanded access, as well as facilitate emergency access for individual patients, as appropriate. Prior experience with respiratory viruses and limited data that have emerged from China suggest that convalescent plasma has the potential to lessen the severity or shorten the length of illness caused by COVID-19.

DONATE PLASMA UPDATE
Today, we are providing an update on one potential treatment called convalescent plasma and encouraging those who have recovered from COVID-19 to donate plasma to help others fight this disease.Ĭonvalescent plasma is an antibody-rich product made from blood donated by people who have recovered from the disease caused by the virus. government, academia and industry to expedite the development and availability of critical medical products to treat this novel virus. Food and Drug Administration has been working with partners across the U.S. Commissioner of Food and Drugs - Food and Drug AdministrationĪs part of the all-of-America approach to fighting the COVID-19 pandemic, the U.S. Women may also consider a double red cell donation or whole blood donation.Stephen M. Female platelet donors may be eligible to donate concurrent plasma, meaning that plasma and platelets are donated at the same time. Platelets are another specialty blood component needed for treatment of hospital patients. Women are encouraged to get their platelet count tested to determine their eligibility for platelet donation. Plasma transfusions that contain these antibodies may lead to complications for recipients, including severe breathing problems and sometimes death. This means that more women carry these antibodies than men. These antibodies develop when an individual is exposed to cells from another person, such as during pregnancy or transfusion.
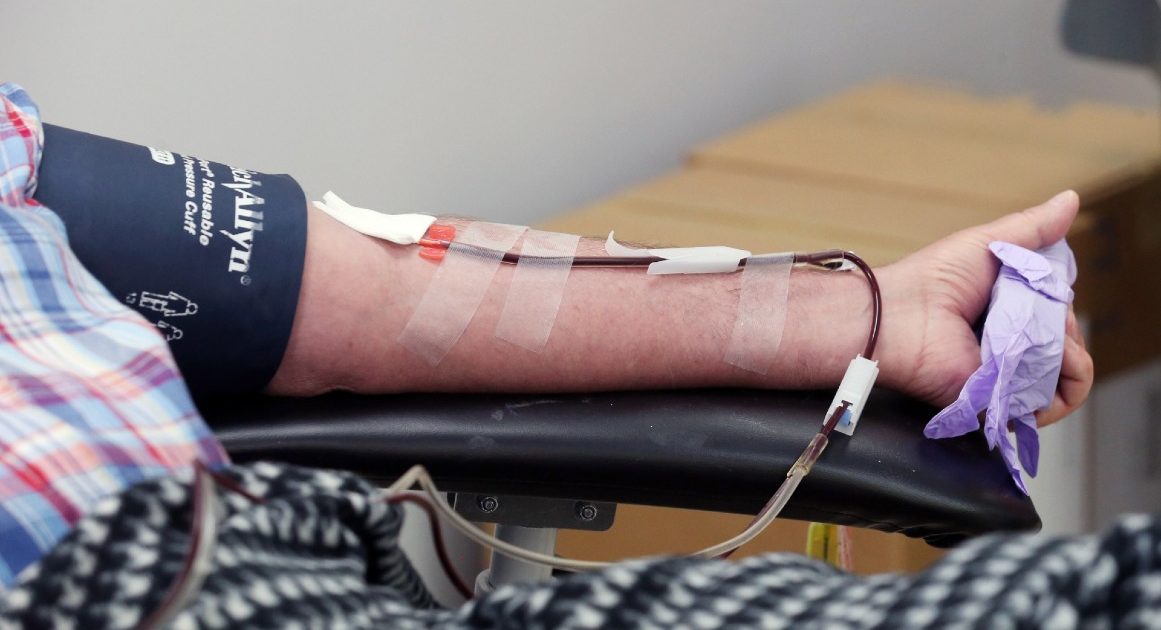
At the Red Cross, those donors are called AB Elite. Type AB donors are always needed to provide plasma for trauma patients.

Individuals can donate plasma as often as every 28 days, and the average donation takes about 1 hour and 15 minutes. Plasma contains antibodies thought to cause TRALI. A plasma donation by type AB donors helps ensure one donor can give even more plasma to help trauma patients. Nebraska Community Blood Bank collects plasma only from males in an effort to eliminate nearly all risk of TRALI (Transfusion-Related Acute Lung Injury)-a rare but serious complication-to plasma recipients.
